Groundbreaking Porcine to Human Cardiac Xenotransplantation: Highlights from a Remarkable Journey
There are over 105,000 Americans currently waiting for organ transplantation1. More than 6,000 of these patients die each year on the transplantation waiting lists1. Xenotransplantation could potentially save thousands of lives but comes with a unique set of risks, including the possibility of triggering deadly immune responses. The first recorded xenotransplantation involved the replacement of a portion of a Russian nobleman's skull in 1682 with a canine graft. Since then, the field saw multiple developmental milestones, with some key ones including xenotransplantation of organ and islet grafts, and subsequent transplantation of genetically modified pig hearts into non-human primates (NHPs). These experiments have all been done with the goal of eventually performing xenotransplantation in human patients, a seemingly distant goal – until now.
The Cardiac Transplantation team at the University of Maryland Medical Center (UMMC) recently performed a groundbreaking transplant of a genetically modified pig heart into a human patient. The team was led by Muhammad Mohiuddin, MD, MBBS, Professor of Surgery and Director of the Cardiac Xenotransplantation program in the Department of Surgery at the University of Maryland School of Medicine and President-Elect of the International Xenotransplantation Association, and Bartley P. Griffith, MD, Professor of Surgery and Director of the Cardiac and Lung Transplant Programs at the University of Maryland School of Medicine. Dr. Mohiuddin has been working in the field of xenotransplantation since 1992. He joined the University of Maryland School of Medicine in 2017 where he and Dr. Griffith founded the first center in the United States for cardiac xenotransplantation research.
The recipient of this genetically modified pig heart is a 57-year-old Maryland resident who arrived at UMMC with long-term, poorly controlled, congestive heart failure. He spent two weeks in the Coronary Care Unit, only rarely able to sit up. During the admission, his condition worsened, and he began to need frequent resuscitation due to recurrent episodes of unstable ventricular tachycardia. At the same time, his ejection fraction dropped to around 10 percent. When medication could no longer support him, he was placed on extracorporeal membrane oxygenation (ECMO) in the Cardiac Surgical Intensive Care Unit for 5 weeks. "We felt that his likelihood of survival was very, very poor, with just continuing his medications in the hospital and leaving him on [ECMO]" says Dr. Griffith. "The intractable arrhythmia barred giving him a [ventricular assist device]."
At the time of the patient's admission, the xenotransplantation team at UMMC had already initiated the approval process with the U.S. Food and Drug Administration (FDA), to transition their promising xenotransplantation research to the human trials phase. The FDA had requested more consistent findings in primates before approving trials in humans. Dr. Mohiuddin and colleagues took this decision seriously and were hard at work to refine and advance their research. However, when faced with a patient who had a strong desire to live, but who was approaching death and had no conventional medical options, Dr. Griffith had the foresight to request emergency authorization of the novel treatment from the FDA under its compassionate use provision. A thorough FDA review of the potential risks and of the particular patient case ensued, putting the diligence and dedication of the UMMC team to the test. The team was up to the challenge. On New Year's Eve, the FDA granted the approvals, allowing the surgery to proceed. Authorization provided for xenotransplantation in this patient alone.
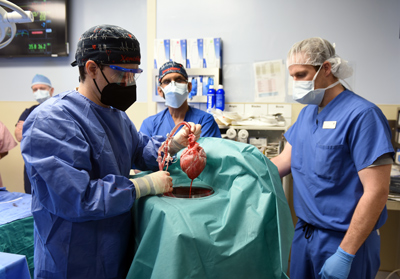
Other hurdles needed to be crossed including insurance limitations, with UMMC and the University of Maryland School of Medicine ultimately agreeing to pick up the bill. The heart xenotransplant was finally performed by the UMMC cardiac transplantation team on January 7, 2022. That morning, the heart was removed from a 1-year-old, 240-pound genetically modified pig supplied by regenerative medicine company Revivicor. The surgical team placed the heart into the XVIVO Heart Box, a perfusion device, to keep the heart preserved until surgery. The operation, although successful, was not without challenges as the surgeons discovered a size mismatch due to the patient's significant cardiomegaly. Dr. Griffith, with more than 30 years of transplantation experience, addressed this, bridging gaps in the vasculature using atrial tissue grafts from the explanted heart. "I took a big breath; I wasn't sure it was going to work," Dr. Griffith says.
Fortunately, the procedure met with success and the heart worked beautifully. The patient was disconnected from life support sooner than expected. Dr. Mohiuddin's laboratory regularly evaluates the patient's heart function to monitor for any signs of rejection. "[He] is progressively improving," says Dr. Griffith. He encountered some "bumps" after his transplant including leukocytosis requiring broadening of his antibiotics but thus far has shown no signs of having acquired a porcine virus from his new heart. The patient will likely remain in the hospital for some three months after his transplant. While his xenotransplant could serve as a bridge to a human heart transplant, Dr. Mohiuddin hopes the new heart will last the rest of his life.
The one-day operation does not do justice to the colossal amount of investigative energy that made it possible. At UMMC alone, Dr. Mohiuddin and Dr. Griffith spent over five years optimizing the process of transplanting pig hearts into NHPs. Leading up to the procedure, the team had to ensure the heart was optimally viable before transplantation, minimize acute injury from preformed anti‐pig antibodies and eliminate any induced clinical or natural immune response when transplanted. Other critical developments leading up to this transplantation include:
- Overcoming microvascular thrombosis caused by immune reaction in primates to carbohydrates in pig hearts. The genetically modified pig from Revivicor had been engineered with deletions of incompatible cell membrane carbohydrates like galactose-alpha-1,3-galactose (Alpha – Gal). Dr. Mohiuddin and colleagues also deleted the Growth Hormone Receptor to reduce the risk of excess growth of the porcine heart.
- The addition of human genes to the genetically modified pig control blood clotting, prevent inflammation and blood vessel damage, and lessen the risk of rejection.
- Utilization of a new perfusion method commercialized by the Swedish company XVIVO for storing and treating the donor heart after it's harvested. The heart was bathed in a special blend of hormones, electrolytes, nutrients, and dissolved cocaine, all of which contribute to minimizing ischemic injury and shown in prior studies to significantly extend xenograft viability.
- Developing the right immunosuppressant regimen to prevent rejection. This included an experimental antibody drug called KPL-404, made by Kiniksa Pharmaceuticals, Ltd. KPL-404 shuts down production of antibodies by binding to CD40 receptors. This monoclonal anti-CD40 antibody was a significant improvement over other promising immune modulators which were associated with coagulation disorders2.
The operation was a major test for several experimental innovations. It may be too soon to say whether pig hearts could lead to what Dr. Griffith calls the "holy grail" -- an unlimited supply of healthy hearts for people who need them. At the very least, the XVIVO system and the drug KPL - 404 could improve the results of conventional organ transplants. The FDA made clear that the one-off experimental surgery will not substitute for a clinical trial. However, with more research and with the FDA's approval, the UMMC surgeon-scientists hope to begin studying porcine-to-human xenotransplantation in a small trial with subsequent plans for a larger multi-center study. It truly is an exciting time to be in the business of organ transplantation.
Referring a Patient
The University of Maryland Transplant Center combines pioneering research such as this historic cardiac xenotransplant with cutting-edge procedures. For example, robot-assisted surgery for nephrectomy and kidney implantation yields expanded options for overweight recipients and donors. UMMC's deep expertise in minimally invasive procedures results in faster healing, less pain, and improved cosmesis.
Call 410-328-5408 to refer a patient. Learn more at umm.edu/transplant.
References:
- Based on OPTN data as of February 14, 2022
- Mohiuddin MM, Singh AK, Corcoran PC, Hoyt RF, Thomas ML 3rd, Lewis BG, Eckhaus M, Dabkowski NL, Belli AJ, Reimann KA, Ayares D, Horvath KA. Role of anti-CD40 antibody-mediated costimulation blockade on non-Gal antibody production and heterotopic cardiac xenograft survival in a GTKO.hCD46Tg pig-to-baboon model. Xenotransplantation. 2014 Jan-Feb;21(1):35-45. doi: 10.1111/xen.12066. Epub 2013 Oct 29. PMID: 24164510; PMCID: PMC5603077.
