Pioneering an Alternative to Open-Heart Surgery for Degenerative Mitral Valve Disease
Researchers at the University of Maryland Heart & Vascular Center have pioneered a beating heart, minimally invasive approach to treating mitral valve posterior leaflet prolapse, potentially giving more patients with mitral regurgitation a surgical treatment option. The approach involved inventing the HARPOONTM Beating-Heart Mitral Valve Repair System (Edwards Lifesciences, Irvine, CA), a device that permits transesophageal echocardiography (TEE)-guided, transventricular chordal repair through a ~5 cm keyhole incision on the chest wall1. A safety and feasibility study of the HARPOON system, which cleared European trials and carries a CE mark, has recently opened in the United States and expects to recruit 360 participants across 40 centers over the next three years.
Mitral Valve Degeneration: A Common Problem That Commonly Goes Surgically Untreated
Mitral valve (MV) regurgitation brought on by degenerative MV disease is more common than aortic valve pathologies, and MV procedures are the fastest-growing area of cardiac intervention. In a retrospective study of 87,214 patients, researchers at the University of Maryland School of Medicine (UMSOM) Valvular Heart Disease Research and Innovation Laboratory discovered that early MV repair restores patients’ survival rates to those of individuals without heart disease. Unfortunately, many patients are referred for surgery too late, after they have already developed comorbidities such as ventricular dysfunction, atrial fibrillation, congestive heart failure and pulmonary hypertension.2
However, conventional MV repair that involves sternotomy or thoracotomy, aortic cross-clamping and cardiopulmonary bypass is associated with high morbidity and mortality for certain people. In fact, the same retrospective study revealed that operative mortality was 3.7 percent for MV replacement and 1.1 percent for MV repair.2 While conventional MV repair remains an excellent treatment approach, outcomes vary depending on surgeon experience, and the residual risks of intervention preclude a significant number of patients with MV regurgitation from receiving treatment.
In a given year, less than 2 percent of patients with severe mitral regurgitation receive surgical treatment for it.3 Low treatment rates are likely attributable to referrer being unaware of minimally invasive options and also because of a paucity of surgeons able to offer high-quality, durable MV repair.
The HARPOON System Enables Minimally Invasive, Beating-Heart, Real-Time MV Repair
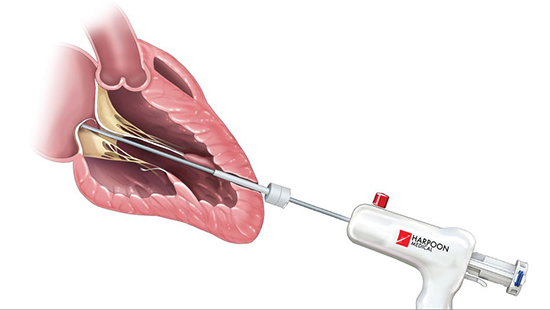
The HARPOON system is designed to be used transapically with TEE-guidance to repair mitral valve posterior leaflet prolapse with self-tying ePTFE chords. Image source: Edwards Lifesciences
The HARPOON system overcomes many of the challenges that limit the number of patients who receive MV repair. Through a low-profile (9 Fr) shaft and guided by TEE on a beating heart, the HARPOON delivery system introduces into the mitral valve preloaded GORE-TEX expanded polytetrafluoroethylene (ePTFE) sutures with double-helix knots that self-tie. (In the experience of the University of Maryland mitral valve repair team, ePTFE neochordal reconstruction gradually and universally replaced leaflet resection for MV repair between 2004 and 20174, a trend also seen at other centers.
Results from a prospective, multicenter European study have demonstrated the following benefits following treatment with the HARPOON system:
- Least Invasive Surgical Approach – The system is used transventricularly, requiring no more than a 5 cm keyhole incision to access the heart, omitting the need for a sternotomy.
- Performed on a Beating Heart – The approach avoids the need for cardiopulmonary bypass, aortic clamping and stopping the heart, avoiding related complications. The surgeon is able to adjust implanted chords in real time, on a beating heart and under TEE-guidance, allowing for titration and optimization of chordal length.
- Less Blood Loss – Between the two clinical trials completed in Europe, only one patient in 62 required a blood transfusion.5,6
- Shorter Operation Times – In the initial trial of the device, operative lengths were about half of those of conventional MV repair.5
One important potential benefit of the HARPOON device is the simplification of MV repair. On average, cardiac surgeons perform 10 MV repairs a year, a volume most find too small to develop confidence in offering the procedure. However, the Harpoon system simplifies some of the most challenging steps in MV repair, making it more easily teachable and adoptable. Moreover, because the approach is so minimally invasive, treatment of mitral valve regurgitation earlier in the disease process may avoid adverse effects on the heart and lungs.
Results of Earlier HARPOON Trials
In recently published data on one-year trial outcomes of 65 patients,6 UMMS and a team of clinical researchers in Poland, United Kingdom and Italy reported that:
- Technical success was achieved in 95 percent of patients treated with the HARPOON system. All patients were alive at discharge, with no stroke or renal failure.
- One year after their procedures, 98 percent of the patients who had achieved technical success had none/trace (52 percent), mild (23 percent) or moderate (23 percent) mitral regurgitation.
- Overall patient survival among those who received the HARPOON procedure was 97 percent at one year; among this group, 98 percent were in NYHA class I or II and 97 percent had still functioning MV repairs.
University of Maryland Heart & Vascular Center’s Critical Mass of Mitral and Tricuspid Valve Trials
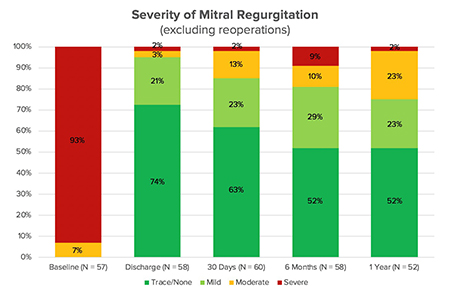
European trials demonstrated promising results for patients treated with the Harpoon Mitral Valve Repair System.
In attempt to gain FDA approval, the HARPOON device is currently under investigation at U.S. centers in a prospective, non-randomized safety and efficacy trial: Beating Heart Mitral Valve Repair with the HARPOON™ System (RESTORE) (NCT04375332). The trial is now open at a handful of centers including the University of Maryland. The UM Heart & Vascular Center is home to a robust clinical trials program, and other mitral and tricuspid valve trials at the center currently recruiting patients include:
- Tendyne Valve – NCT03433274: Clinical Trial to Evaluate the Safety and Effectiveness of Using the Tendyne Mitral Valve System for the Treatment of Symptomatic Mitral Regurgitation (SUMMIT)
- TMVR – NCT03242642: Transcatheter Mitral Valve Replacement with the Medtronic Intrepid™ TMVR System in Patients with Severe Symptomatic Mitral Regurgitation (APOLLO)
- EVOQUE Valve – NCT04221490: Edwards Transcatheter Tricuspid Valve Replacement: Investigation of Safety and Clinical Efficacy Using a Novel Device (2019-06 TRISCEND Study)
For more information about cardiac surgery at the University of Maryland Heart & Vascular Center, call 410-328-5842. To inquire about clinical trials at the center, contact Manal Al-Suqi at 410-328-9409.
Disclosures
Studies of the HARPOON device are being funded by Edwards Lifesciences. At the present time, the U.S. Food and Drug Administration has not approved the HARPOON device for patients in the United States.
Related Content
- UMMC Physician Brief – Benefits of Evolution to a Simpler Mitral Valve Repair Technique
- Clinical Trial (NCT02432196) – CE Mark Study for the Harpoon Medical Device in Poland
- Clinical Trial (NCT02768870) – CE Mark Study for the Harpoon Medical Device (TRACER)
1Gammie J, Patel R, Ghoreishi M, inventors; University of Maryland, Baltimore, assignee. Transapical mitral valve repair device. US patent 8,852,213B2. October 7, 2014.
2Gammie JS, Chikwe J, Badhawr V, Thibault DP, et al. Isolated mitral valve surgery: the Society of Thoracic Surgeons adult cardiac surgery database analysis. Ann Thorac Surg. 2018;106(3):716-727. doi: 10.1016/j.athoracsur.2018.03.086
3Head SJ, van Leeuwen WJ, Van Mieghem NM, Kappetein AP. Surgical or transcatheter mitral valve intervention: complex disease requires complex decisions. EuroIntervention. 2014;9(10):1133-5. doi: 10.4244/EIJV9I10A191
4Parsrija C, Tran D, Ghoreishi M, Kotloff E. Degenerative mitral valve repair simplified: an evolution to universal artificial cordal repair. Ann Thorac Surg. 2020;110(2):464-473. doi: 10.1016/jathoracsurg.2019.10.068
5Gammie JS, Krzysztof B, Gackowski A, D’Ambra MN, et al. Beating-heart mitral valve repair using a novel ePTFE cordal implantation device: a prospective trial. J Am Coll Cardiol. 2018;71(1):25-36. doi: 10.1016/j.jacc.2017.10.062
6Gammie JS, Krzysztof B, Gackowski A, Piotr Szymanski, et al. Safety and performance of a novel transventricular beating heart mitral valve repair system: 1-year outcomes. Eur J Cardiothorac Surg. 2020;ezaa256. Online ahead of print. doi: 10.1093/ejcts/ezaa256
